Like most of the BioStrata team, I still keep an eye on current research trends, both on behalf of our clients and just out of general interest. Of course, this is much easier to do when a recent paper of interest has your name on it! The paper in question is about how cells take in factors from their environment and was just published in Science - a story so interesting, I wanted to share it with you all!
“Let me in!”
A few years ago, I joined one of the leading research labs in the field of structural biology and cellular transport, to team up with Dr Kelly and Prof. Owen on a mission to understand the finer points of how cells transport necessary material within and between themselves. Let me explain a little more.
Cells are protected from their surroundings via their cell membrane. However, they need to interact with the environment in order to be able to take up nutrients, regulate their growth and many other complex cellular processes. Many such processes involve the transport of molecules across this membrane or the addition, removal or modification of different types of molecules within the membrane such as specific lipids or receptors. Like most biological processes, these are tightly regulated.
The link between cellular transport, getting ill and treating disease
The variety and complexity of the transportation routes across the membrane are especially fascinating, in particular because many of them are exploited by pathogens (e.g. influenza virus). One of the best understood routes into most cells employs clathrin, a scaffold molecule that helps deform a small area of the membrane, bending it inwards, to then be pinched off as little bubbles, known as vesicles. Cells rely upon vesicles to deliver nutrients and other selected cargo from the environment surrounding the cell. They may also contain pathogens, such as viruses, which disguise themselves as cargo. Researchers aim to explore this pathway into the cell for effective and selective drug delivery or gene therapy.
However, clathrin lacks the ability to bind membranes on its own and relies on adaptors as a physical bridge connecting it to the membrane and the cargo. These adaptor molecules are vital for aiding clathrin to form a cage at the right site, at the right time. The assembling clathrin cage bends the membrane locally and stabilises the bend membrane as the vesicle forms. The most common adaptor, Assembly Polypeptide 2 (AP2), is capable of binding many types of specific cargo molecules and works alongside clathrin to transport this cargo into the cell.
It is well established that AP2 is the key molecule recruiting clathrin to the cell membrane and linking it to cargo and membrane. In the absence of AP2, scientists could not find any clathrin-coated vesicles at the cell membrane, demonstrating that AP2 is an essential component in the process. However, it was puzzling why AP2 wasn’t allowing clathrin cage formation at a membrane in a cell-free experiment, where no cells are used but only isolated molecules. This finding could mean that the presence of AP2 may either not be sufficient for clathrin cages to start forming, or that clathrin cage formation only occurs under certain circumstances that control the accessibility of AP2.
In addition, we also wanted to find out how cells ensured that clathrin cages did not form spontaneously upon “bumping” into adaptors but only in the presence of cargo and at the right location. Using a specific part of the AP2 molecule that is still able to bind both the membrane and clathrin, we could demonstrate clathrin cage formation in the presence as well as absence of membrane. This was a hint that the whole AP2 molecule should be able to drive clathrin cage formation under certain circumstances. Therefore we needed to demonstrate in a cell-free experiment that isolated AP2 as a whole is able to drive clathrin cage formation in the presence of membrane and cargo and in the absence of any other molecule.
Months of work (time well spent)
After months of hard work and a very successful collaboration with three other labs (Prof Ungewickell and Dr Dannhauser, Prof Höning and Dr Graham) we achieved exactly that – clathrin-driven vesicle formation enabled by membrane and cargo bound AP2 in the absence of any other molecule. However, proving this idea wasn’t all that we achieved. We also found that AP2 can switch between two states, one of which prevents tight clathrin binding. This inactive state is switched to an active state by a change within the AP2 molecule that is initiated by simultaneous binding of AP2 to both membrane and cargo, which then allows efficient clathrin binding and cage formation. This switch mechanism therefore prevents clathrin from binding to AP2 and forming transport vesicles when no cargo is ready to be taken in.
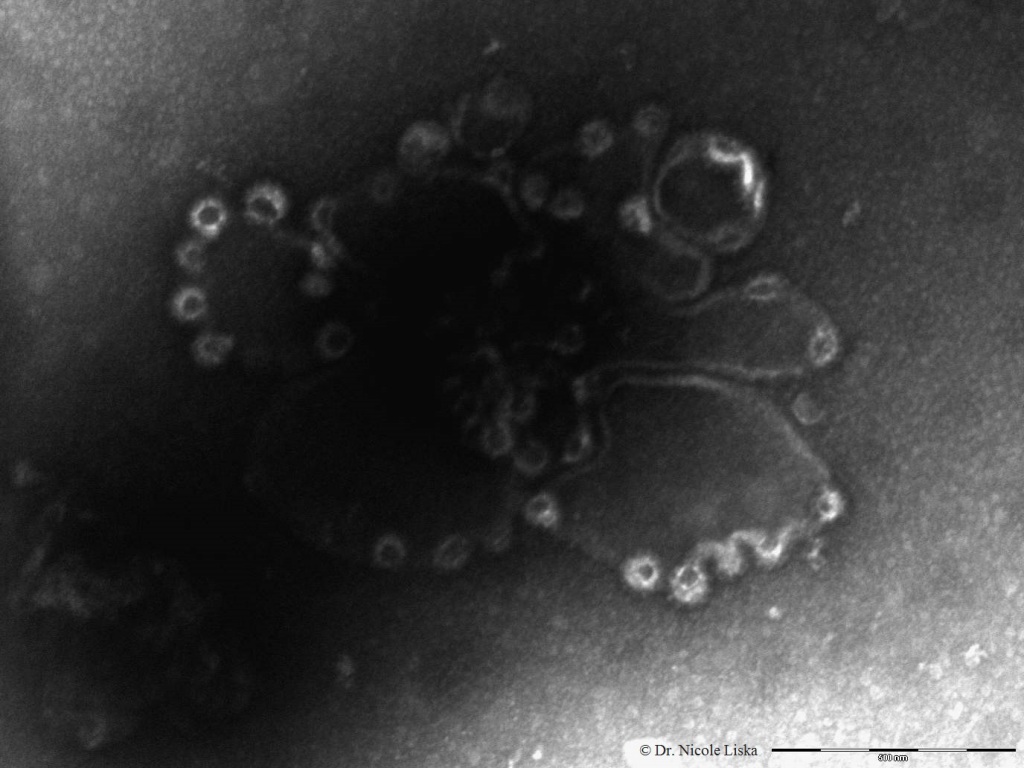
 This scanning electron microscope (SEM) image shows the clathrin cages (bright circular structures at the edges) assembling on an artificial cell membrane. The scale bar is 500nm.
This scanning electron microscope (SEM) image shows the clathrin cages (bright circular structures at the edges) assembling on an artificial cell membrane. The scale bar is 500nm.Why is this research important?
This finding is indeed a great advancement for the understanding of how selected cargo molecules are transported into the cell, providing detailed knowledge of the role of an essential component involved in this mechanism.
This mechanistic insight can serve a much wider area of research including the development of biomolecular drugs designed to only activate at a certain site or when in contact with a specific combination of molecules. Also, this system could be used for efficient membrane encapsulation of different types of biomolecules such as DNA (useful for applications such as gene therapy).
Want to learn more?
Are you keen to learn more about clathrin and cellular transport? Well, you can find the original paper published in Science (July 2014) here, which includes all the mechanistic and structural details of this study. Have fun!